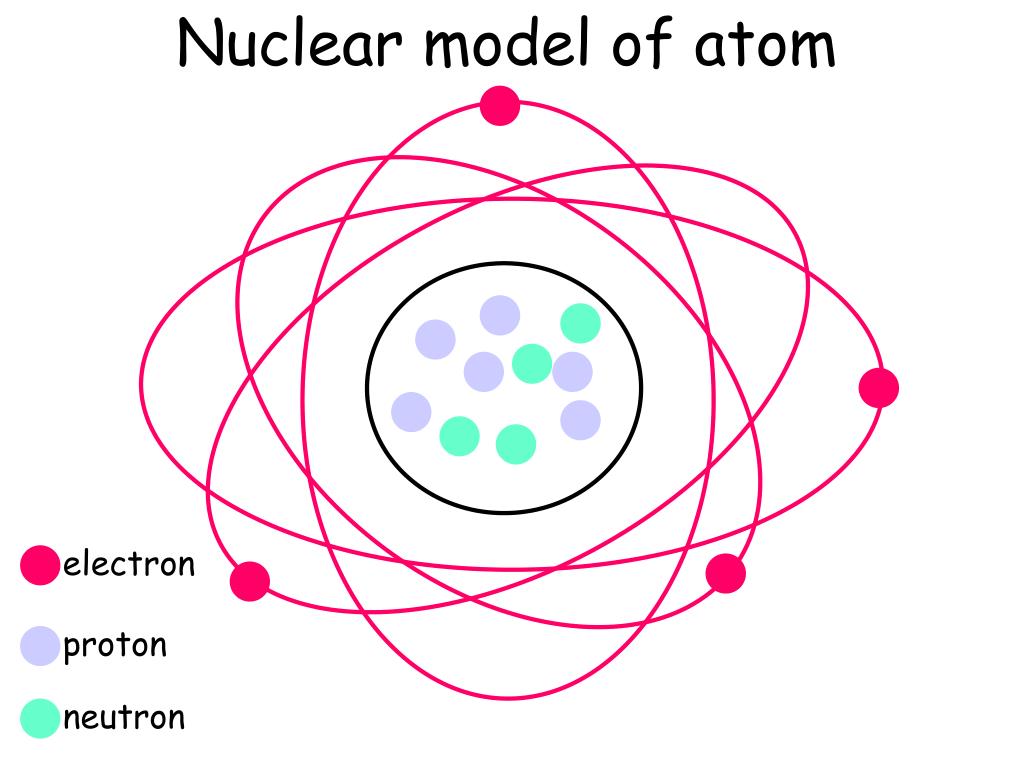
Deuterium is stable and makes up 0.0156% of naturally occurring hydrogen and is used in industrial processes like nuclear reactors and Nuclear Magnetic Resonance. Protium is stable and makes up 99.985% of naturally occurring hydrogen atoms.ĭeuterium contains one neutron and one proton in its nucleus. The most abundant isotope, hydrogen-1, protium, or light hydrogen, contains no neutrons and is simply a proton and an electron. Mass numbers of typical isotopes of Hydrogen are 1 2. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. 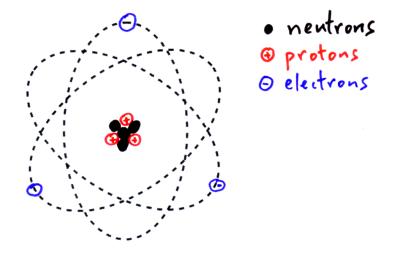
The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. Hydrogen is a chemical element with atomic number 1 which means there are 1 protons in its nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
